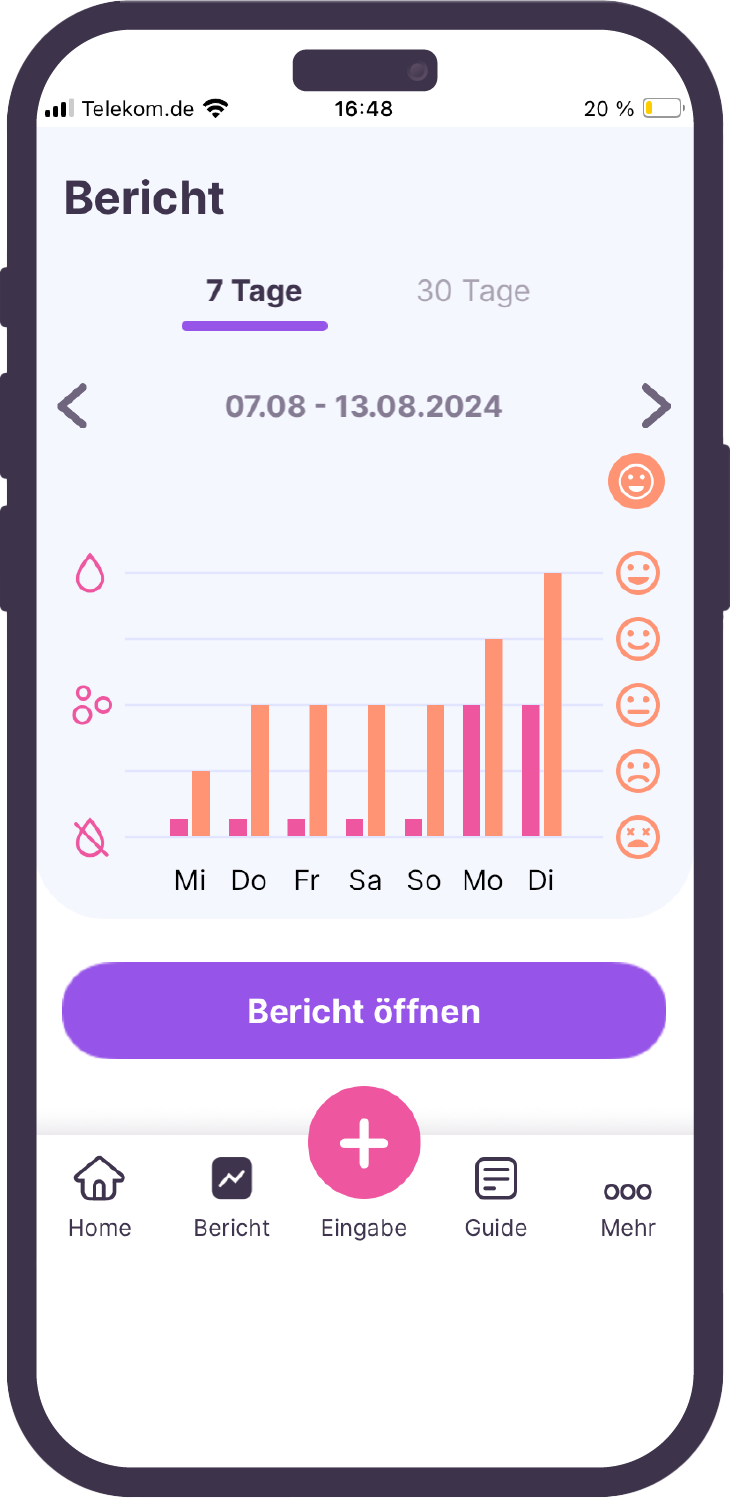
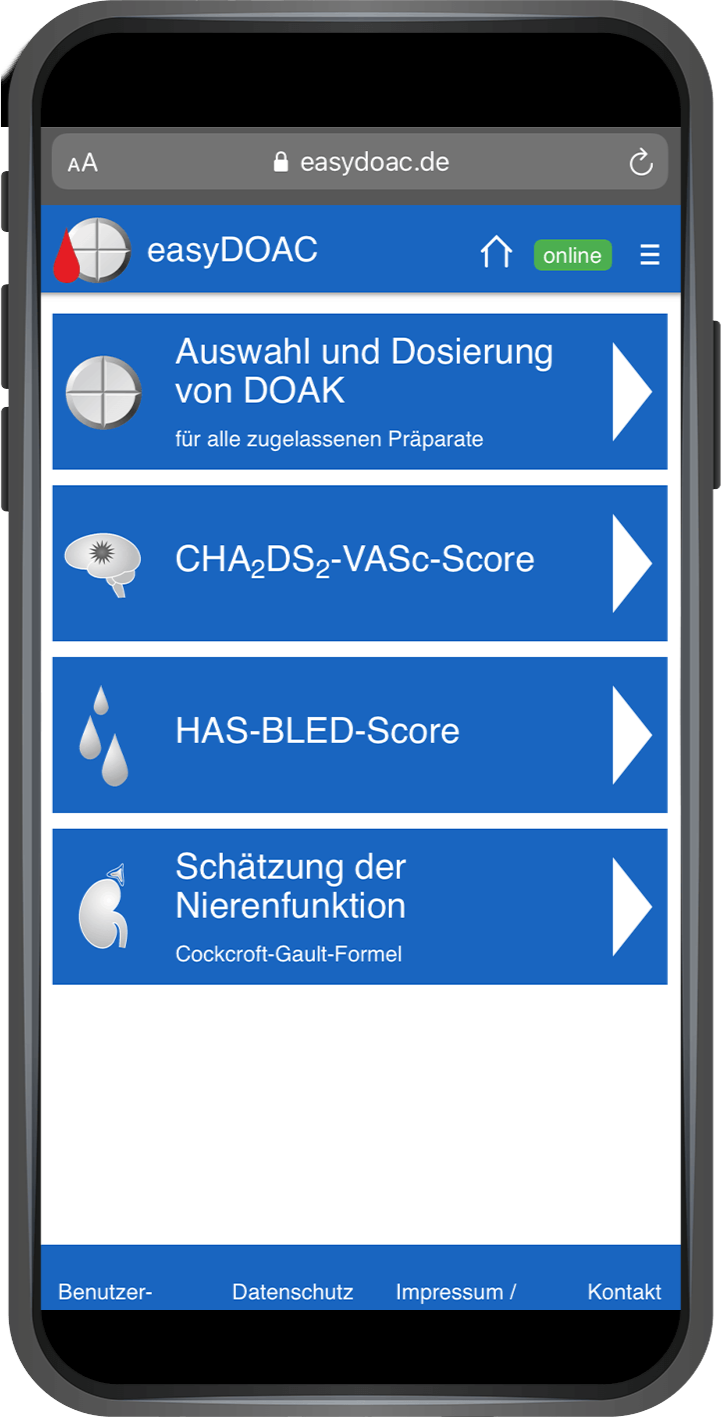
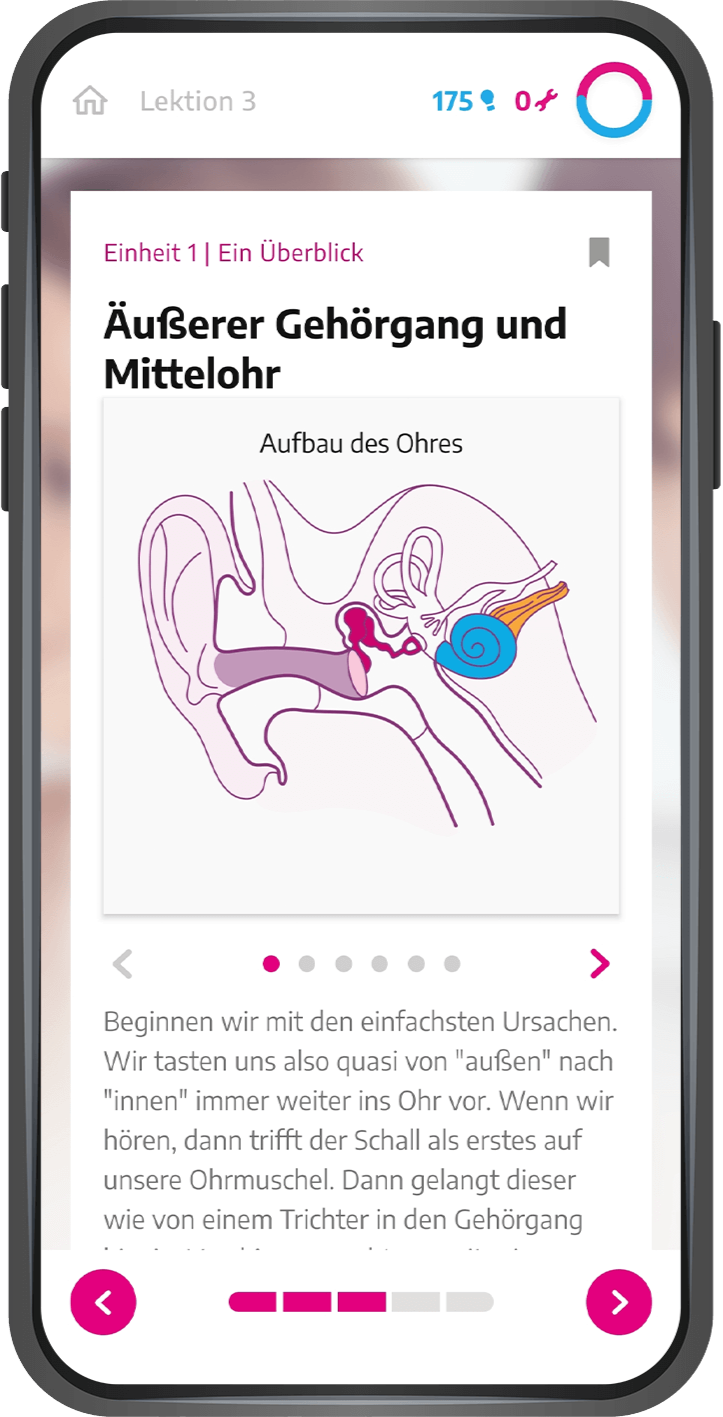
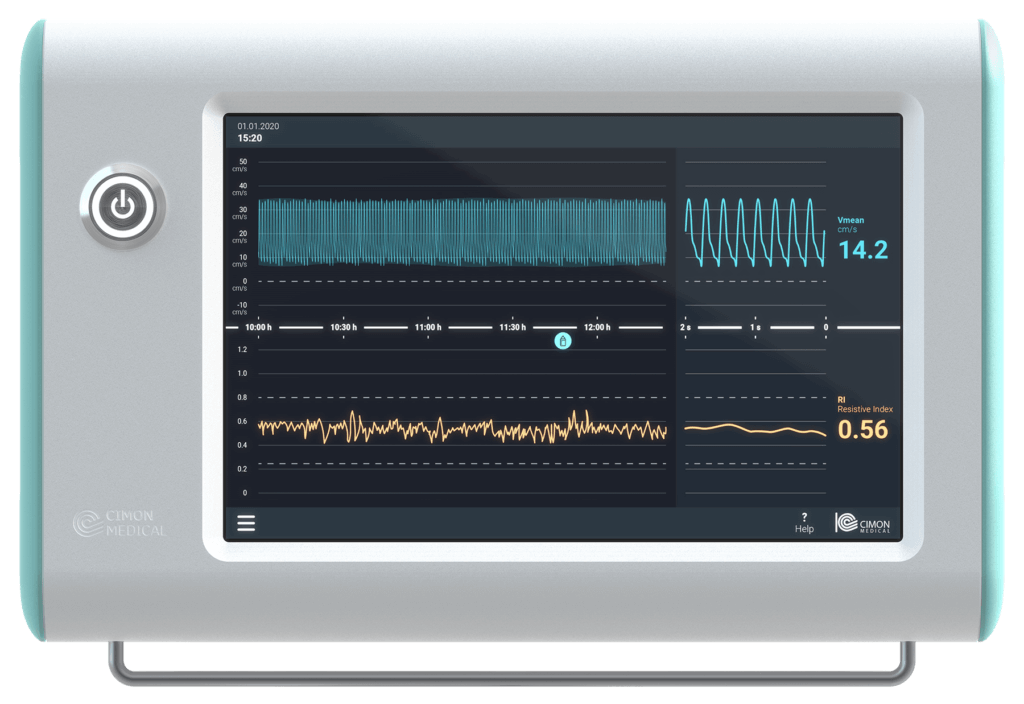
Our services range from regulatory affairs and quality assurance measures to post-market surveillance. We can always draw on the expertise of the BAYOOMED business unit and the associated experience in medical device classes I, IIa, IIb and III as well as software safety classes A, B and C .