
Legal Manufacturer as a Service: Your legal manufacturer for medical devices in over 30 countries
As an experienced legal manufacturer, BAYOOCARE assumes full manufacturer responsibility for your medical device – from classification and CE marking to post-market surveillance. You concentrate on development and marketing, we take overall regulatory responsibility.

Who we are
Regulatory experts with practical experience: BAYOOCARE is a company within the BAYOONET Group that specializes in medical devices. For over ten years, we have been supporting medtech companies, pharmaceutical companies and start-ups in the approval and marketing of medical devices and in-vitro diagnostics. Our team combines regulatory know-how, clinical expertise and practical project experience – in an area that leaves little room for error.
Responsibility as a service: What sets us apart from traditional consultants: We not only provide consulting services, but also bear full regulatory responsibility for your product as a certified legal manufacturer. With our MDSAP and ISO 13485 certification from TÜV Süd and ISO 27001 certification, we are equipped for all relevant markets. Our EU Regulation 2017/745 certificates for Class IIa and Class IIb prove that we live up to this responsibility every day.
This is how we work
Regulatory Affairs is not an end in itself. We think in terms of your product: What does it need to get onto the market? What does it need to stay there? We are not consultants at a distance, but a reliable part of your team. Pragmatic, because we know that time is a scarce resource in MedTech development. Personally, because regulatory responsibility requires trust. And holistic, because compliance is not a project, but an ongoing process.

Still have questions? Please contact uswe know the answer.
Our MDSAP and ISO 13485:2016 certification by TÜV Süd offers you strategic advantages for international expansion. The Medical Device Single Audit Program enables simplified market approval in the five MDSAP countries: Australia, Brazil, Canada, Japan and USA. We cover the requirements of these important markets with a single audit – saving time and audit effort.

MDSAP builds on ISO 13485 and expands it to include authority-specific requirements. The FDA, ANVISA (Brazil) and MHLW (Japan) accept MDSAP audit reports instead of their own audits. The Australian TGA accepts the MDSAP certificate as proof of ISO 13485, and Health Canada uses it to maintain its Canadian license.
Our ISO 27001 certification proves that we systematically manage information security – centrally for digital health applications and software medical devices that process personal health data. We ensure that your technical documentation meets all requirements for GDPR, data security and the basic security and performance requirements.
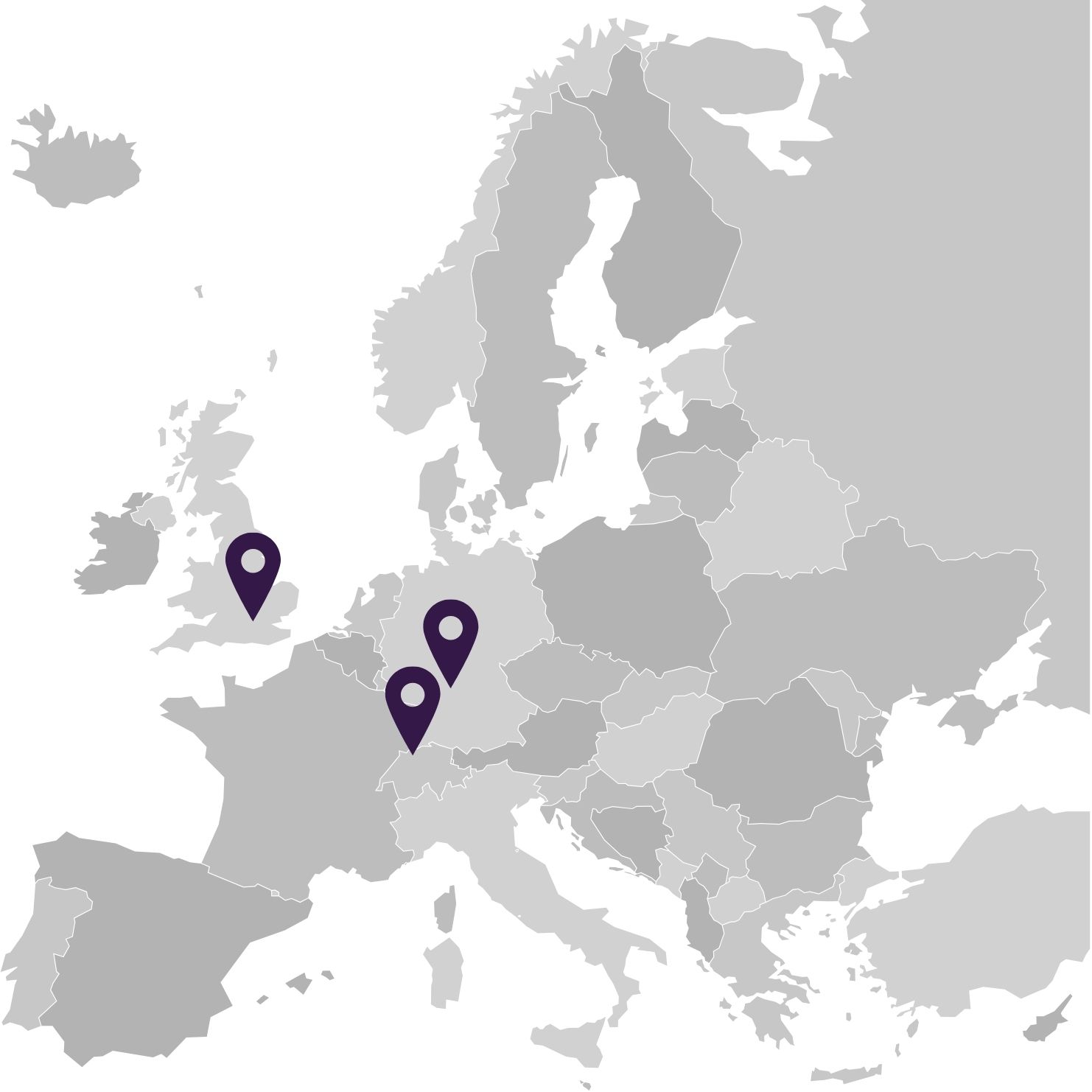
With our locations in Germany, Switzerland and the UK, we are ideally positioned for your international expansion. Thanks to our local presence, we are familiar with the specific requirements and regulatory peculiarities of the respective markets and can provide you with targeted support for your approval projects.
International market approval raises many questions. We have answered the most common ones for you – from the role of the legal manufacturer to MDSAP certification and country-specific requirements. Your question is not listed? Please feel free to contact us.


